Myopia control prevents the progression of myopia through treatments that slow down the lengthening of the eye in children and teens. Through myopia control, patients may also benefit from reduced risk of developing conditions such as cataract development, glaucoma, retinal detachment and macular degeneration.
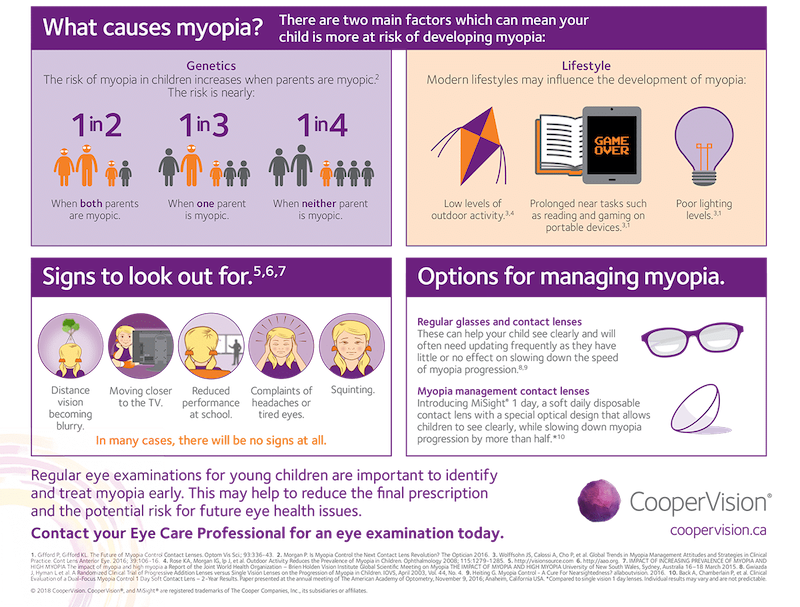
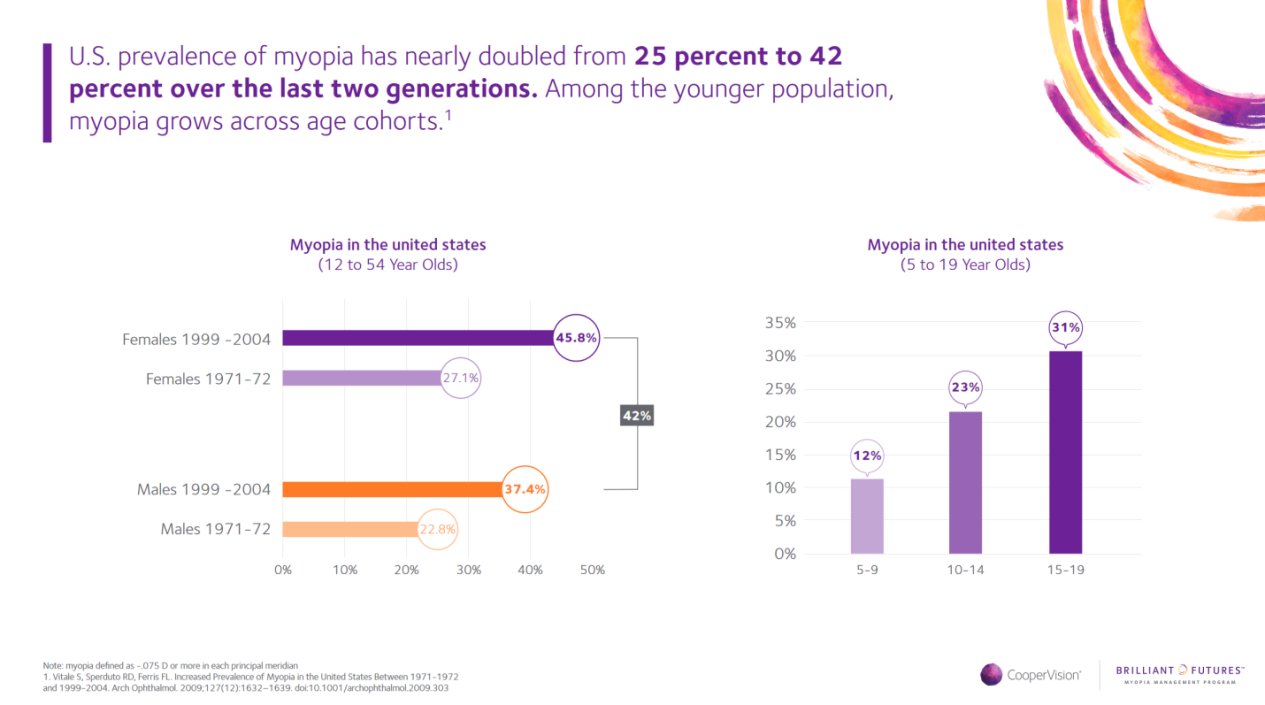
When it comes to the prevalence of myopia, the statistics are staggering. In the U.S. alone, more than 40 percent of Americans have myopia and that number is increasing at an alarming rate, especially among school-aged children.1 CooperVision and Eye Care and Wear is helping to slow the upward trend in severity through its Brilliant Futures™ Myopia Management Program featuring MiSight® 1 day contact lenses, the first and only FDA approved* contact lens to slow myopia progression of myopia in children (aged 8-12 at the initiation of treatment). A recent clinical study reveals that MiSight® 1 day reduced the rate of myopia progression in that age group on average by 59% over a three-year period.2†
Call our office to learn more about Myopia Control Soft Conact Lenses
UPPER MONTCLAIR (973) 707-5255
RIDGEWOOD (201) 652-8080

Infographic about Myopia stats
1 Cooper, Y. (2019, May 1). With Childhood Myopia Rates on the Rise, the American Optometric Association Highlights the Importance of Early Intervention through Annual Eye Exams. Retrieved from https://www.aoa.org/newsroom/myopia-rates-on-the- risesyvm
*MiSight® 1 day (omafilcon A) soft (hydrophilic) contact lenses for daily wear are indicated for the correction of myopic ametropia and for slowing the progression of myopia in children with non-diseased eyes, who at the initiation of treatment are 8-12 years of age and have a refraction of -0.75 to 4.00 diopters (spherical equivalent) with ≤ 0.75 diopters of astigmatism. The lens is to be discarded after each removal.
†Compared to a single vision 1 day lens over a 3 year period.
2 Chamberlain P, et al. A 3-year randomized clinical trial of MiSight® lenses for myopia control. Optom Vis Sci. 2019; 96(8):556-67




